

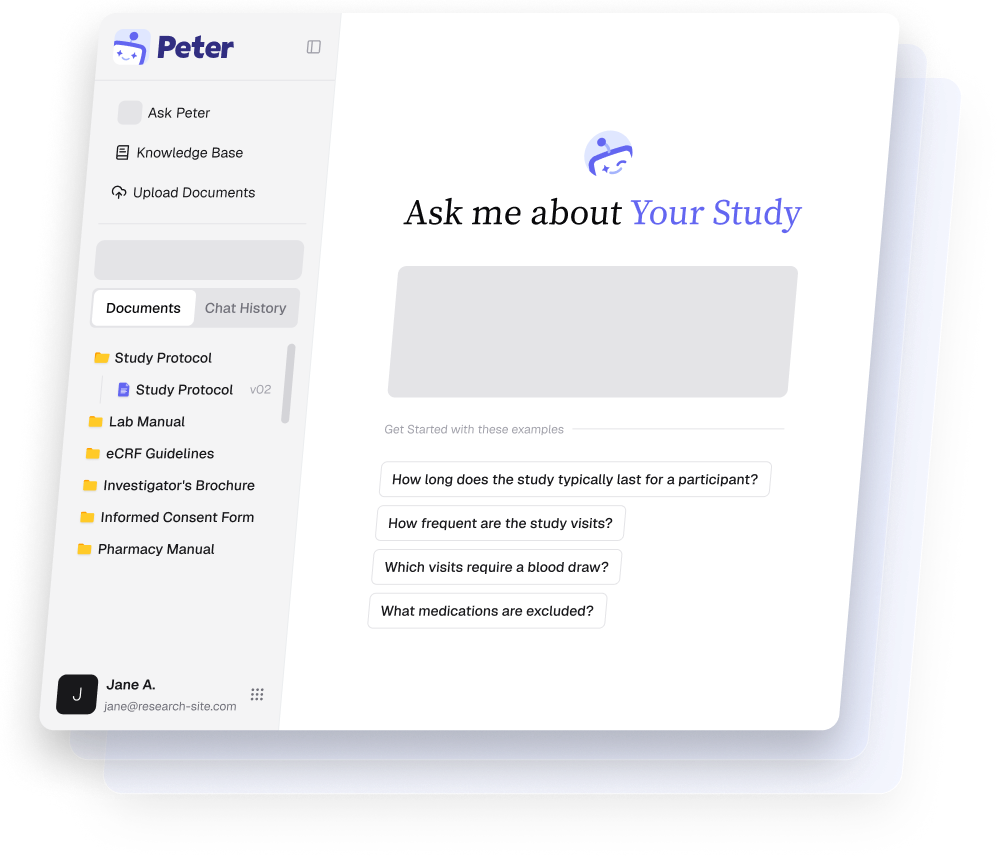
Find answers from
study documents.
Helping sites deliver high-quality research with confidence.


Helping sites deliver high-quality research with confidence.


Protocols and manuals are long. Key details are scattered. Ctrl+F only works when you already know the exact document and phrase.


Provide a summary of the study, including key inclusion/exclusion criteria for prescreening.
Is prednisone prohibited or restricted?
Are there special instructions that study participants should be aware of prior to study visit 2?
If a participant missed two doses of the investigational product, can treatment be resumed per the protocol?
What are the processing and shipping requirements for PK samples?
I have a monitoring visit — can you give me pointers to help me prepare?
What is the reporting timeline for suspected unexpected serious adverse reactions (SUSARs)?
What steps should I follow to complete study closeout?


Peter is most helpful for verifying prohibited concomitant medications and exclusion criteria, especially when the lists are extensive.
Principal Investigator

Like the mini protocols, Peter puts sponsor study documents at my fingertips. But, Peter helps me find information I need within seconds. This is revolutionary.
Clinical Research Coordinator

New study onboarding is stressful. Quick access to the right info makes screening and randomization much more efficient and helps prevent mistakes.
Clinical Research Coordinator